What could those 2 possibly have in common? Both could be causing us to eat more. The mechanism and the reason for each is very different though. Let’s have a look.
Fructose
In a recent podcast, Peter Attia introduced professor Rick Johnson who is an expert in fructose metabolism. He revealed that fructose is the only substance known so far that gets metabolized fully. Meaning that normally a cell will have a feedback mechanism when cellular energy (ATP) goes low. With fructose however, the cell doesn’t stop and ATP can get really low. The details were not discussed but this would have the effect to signal to the rest of the body that there is an energy crisis. As a consequence, hunger is stimulated and overall metabolism is reduced. A perfect scenario to gain weight. The effect is largely depending on 2 factors and that is dose and speed. If either one increases then it triggers this effect. Did you enjoy a tasty orange juice this morning?
That is not all what was revealed in the podcast but I suggest you head on over and listen in as it is bursting with interesting info. Kudos to Peter Attia for bringing this guy to the front.
To reveal a bit of the detail behind this, it is the fructose phosphorylation by fructose kinase in the liver for which there is no feedback (1). The phosphorylation happens through ATP donating one phosphor atom so you get fructose enriched with one phosphor.

With no feedback, you can see how taking in a bigger bolus of fructose can decrease ATP sufficiently to get the cell stressed out through too low ATP levels.
Continuous high fructose consumption looks detrimental and a key driver to insulin resistance, diabetes etc.
If you are developing cancer at the early stages or beyond, it will happily live on fructose (2) as much as it can on glucose. No wonder we find it associated with cancer (3). It is even less surprising if we know that fructose can cause vasoconstriction (4).
Vasoconstriction goes in hand with hypoperfusion which can lead to tissue hypoxia (5), a central regulator of the hallmarks of cancer. Of course I’m not talking about hypoperfusion at the levels you would find it in the emergency room. Just that lower than optimal level, a suppressing effect that has a very gradual effect over a long term. Speculation I know but given the physiological effects it is very plausible.
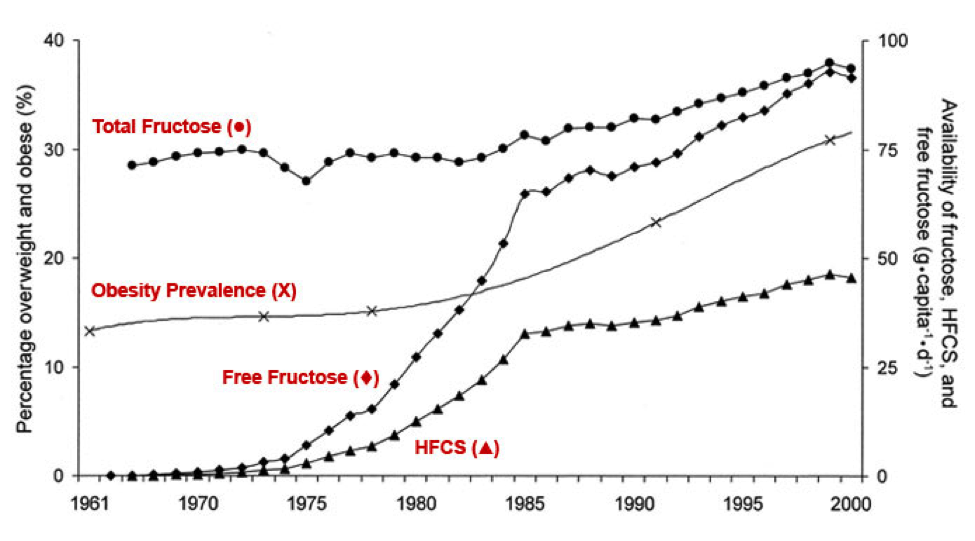
I’ve written about oxygen and CO2 before (6)(7) and how glucose may lead to a slightly lower oxygen saturation in the tissue. With fructose on top, I’m further convinced this lack of sufficient oxygen is taking place in our bodies with our standard diet today. Be it high fructose corn syrup or just plain sugar, they all have roughly an equal amount of glucose and fructose.
Resources
(1) “The negative and detrimental effects of high fructose on the liver, with special reference to metabolic disorders” – Brandon H Mai and Liang-Jun Yan – 2019 – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6549781/
(2) “Increased utilization of fructose has a positive effect on the development of breast cancer” – Xiajing Fan, Hongru Liu, Miao Liu, Yuanyuan Wang, Li Qiu and Yanfen Cui – 2017 – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5622605/
(3″) “The role of fructose in metabolism and cancer” – Charrez B, Qiao L, Hebbard L – 2015 – https://www.ncbi.nlm.nih.gov/pubmed/25965509
(4) “The mechanisms underlying fructose-induced hypertension: a review.” – Klein AV, Kiat H – 2015 – https://www.ncbi.nlm.nih.gov/pubmed/25715094
(5) “Choice of Pharmacological Agents in Hypoperfusion Syndrome” – J.-L. VincentE. Silva – https://rd.springer.com/chapter/10.1007/978-88-470-2296-6_16
(6) “Ketones and oxygen” – https://designedbynature.design.blog/2019/12/12/ketones-and-oxygen/
(7) “CO2 and nutrition” – https://designedbynature.design.blog/2019/12/03/co2-and-nutrition/
Protein
The effect is very different for protein. It is hard to proof so consider this simply for what it is, a hypothesis. But I suspect that there is a protection mechanism to the amount of protein that is needed by the body. Your body may sense how much is available for protein synthesis and will trigger hunger to satisfy that need if it is not met. I have a number of indications and some research pointing out the mechanism.
First lets have a look at a recent mouse study where they wanted to investigate the carb-insulin hypothesis. I’m not interested in the hypothesis itself but they tested a large variety of macro compositions (fat, protein, carbs). Either fat or protein was fixed and when it was fixed they tested both a high amount and a low amount of the fixed component.
Before getting into the study, you have to know that mice on a ketogenic diet, when they want to keep the mice at equal weight, have to be fed about 12% extra. They burn through that extra energy by generating more heat. A pure waist of energy, why?
Protein within the mice diet has to be kept very low because they would otherwise convert too much protein into glucose and that would hinder ketogenesis. Already here I would ask the question: could it be that the mice try to burn so much energy so that they could eat more, thereby getting their needed daily amount of protein?
Now, let’s look at the result of the study (1). There are a number of noticeable effects.

Energy intake under fixed low protein is higher than under fixed high protein. Is this to reach sufficient protein?
Under fixed high fat, shifting from low carb to high carb reduces the protein intake. This leads to a higher energy intake. Again, are we eating more to get to those protein?
Fixed high fat versus fixed low fat, there is a greater energy intake under high fat. Fat does not produce as much glucose. Could more glucose be sparing protein conversion to glucose? And high fat intake automatically means lower protein intake versus the low fat. Could it be a combination of higher glucose intake sparing protein conversion to glucose and the low fat allows more protein intake?
What carb intake does is stimulate insulin. Insulin will oppose glucagon, preventing its production so that less amino acids are converted into glucose.
But insulin is not a fix-all. Under fixed high fat we see a drop in lean mass as we get to the higher end of carb intake. The protective effect of insulin is not sufficient to support a very low intake of protein, still leading to muscle catabolism.
We see the same is true for the fixed low fat, as carbs increase, protein reduces in the food.
What this all looks like is that there is a certain level of amino acid availability needed. There are several ways to obtain this level.
- Through dietary intake but if your food is low in protein then you need to eat more. In order to be able to eat more, you have to be able to burn off the excess energy.
- Through preventing conversion of amino acids to glucose. This can be achieved by taking in more glucose due to the insulin effect.
When we think of humans and look at the ketogenic diet, we do not have to be in the problematic zone of insufficient protein. If you produce high enough ketones, your protein demand may even go down. Ketones are both protecting the skeletal muscle from catabolism and also lower glucose output from the liver. Likely, the more full your liver is with glycogen, the more it can release through the action of glucagon. In such case insulin has to rise a little bit to counter that effect to keep plasma glucose nicely under control.
What applies to the mice, could that be appliceable to humans? Statistics don’t prove everything but let’s look at one anyway to get a hint of it.

Across time we see that the quantity of protein consumed remains more or less the same yet carbohydrate and certainly fat content increases. Purely speculating on the slight increase in protein… this may be because we get more obese which results in larger skin mass and cells which all require protein to maintain them.
On a standard diet, all these macros are combined in our meals. What this shows is that our meals are containing less protein in % of total caloric intake. So could it be that, because our meals are containing less protein, we naturally start to eat more in order to meet the protein need? Just like the mice seem to do?
If this is true then you could argue for prioritizing protein to meet your needs. This is essentially what the Protein Leverage hypothesis is about. Don’t think that this is just a vague idea though. There is extensive research that looked into this using animal studies. A review paper (a summary of research on a specific topic) looking at the effect in the brain concluded that feeding behavior is influenced by its amino acid sensing (2).
It doesn’t answer the question how much protein you should but you don’t have to know. Your body tell you!
I’m still curious though, how are the mice on a ketogenic diet able to upregulate their heat production, assuming this is in response to the low protein intake? Or is that a coincidence and purely ascribed to ketones which do cause the uncoupled metabolism in the mitochondria (through UCP1) (3).
Resources
(1) “The carbohydrate-insulin model does not explain the impact of varying dietary macronutrients on the body weight and adiposity of mice” – Sumei Hu, Lu Wang, Jacques Togo, Dengbao Yang, Yanchao Xu, Yingga Wu, Alex Douglas, John R.Speakman – 2020 – https://www.sciencedirect.com/science/article/pii/S2212877819309421
(2) “Central Amino Acid Sensing in the Control of Feeding Behavior” – Nicholas Heeley and Clemence Blouet – 2016 – https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5120084/
(3) “Mitochondrial biogenesis and increased uncoupling protein 1 in brown adipose tissue of mice fed a ketone ester diet.” – Srivastava S, Kashiwaya Y, King MT, Baxa U, Tam J, Niu G, Chen X, Clarke K, Veech RL – 2012 – https://www.ncbi.nlm.nih.gov/pubmed/22362892
Leave a comment