Conversion of protein to glucose
Among the keto crowd we have people who go full carnivore. Especially in the carnivore community they have posed that the conversion of protein to glucose (gluconeogenesis or GNG) is demand driven so excess amino acids (from the protein eaten) do not convert to glucose if there is no demand for it.
For example Amber O’Hearn has written about this in 2012: http://www.ketotic.org/2012/08/if-you-eat-excess-protein-does-it-turn.html
Studies such as the following seem to further support the idea.
“Dietary Proteins Contribute Little to Glucose Production, Even Under Optimal Gluconeogenic Conditions in Healthy Humans”, Claire Fromentin, Daniel Tomé, Françoise Nau, Laurent Flet, Catherine Luengo, Dalila Azzout-Marniche, Pascal Sanders, Gilles Fromentin, and Claire Gaudichon, 2013, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3636601/
However they are not complete by only looking at blood glucose that appears from the tracer amino acids. It does not account for any possible GNG in the liver which could be converted to storage as glycogen. And when it comes to the question of supply vs demand, it needs to take into account the hormonal response noted by glucagon and insulin.
Glucagon does cause the breakdown of glycogen (glycogenolysis) from the liver so it is reasonable to think there is no glycogen refill during stimulation of breakdown.
The clarity on this demand/supply question is important for the keto-carnivore and anyone who eats a high protein diet. By that I’m not saying eating carnivore means eating a high protein diet but there are people who do just that.
The reason it could be of importance, for those who care, is that if there is a supply driven conversion of excess dietary protein to glucose then you risk going out of ketosis. Whether this is good or bad is not the point of this article. What I want to do here is look at available knowledge and help those who are in this situation of wanting ketone production, to evaluate whether it is demand or supply driven.
You’ll want to adjust your protein intake to avoid stepping too much out of a ketogenic state if that is what you are looking for. That is, if GNG is supply driven.
The level of glycogen in the liver is a determining factor for getting into and out of ketosis. So we need to look into this and see if there is any effect on the glycogen storage.
For the majority, the mindset in the community is already determined that it is demand driven so I’ll take the stance of trying to disprove it. Full disclosure, I have previously written about this, taking that same stance. I have in the meantime found a few more relevant things which I’ll discuss in this article.
Previous post: https://www.reddit.com/r/ketoscience/comments/dw76n5/dietary_protein_and_glucose/
I think I’m able to further convince due to a number of points that are missed in the demand driven arguments. These points below come on top of what I have already written down.
- intravenous (IV) studies versus actual dietary protein intake & cortisol
- kidney GNG
- longevity
- Type 1 diabetes
Apart from those points I also don’t see any clarity on where the excess protein end up if it is demand driven. I have not seen any material that shows there is a pool of amino acids somewhere in the body that is refilled with the excess protein intake. If there is no such pool then what happens to the excess? All used for extra protein synthesis? There is also no data showing that protein synthesis is linear to amino acid availability indefinitely.
These are important questions that need an answer when GNG is demand driven.
But OK, let’s start with the way tests are done.
Real life studies vs IV
There’s a couple of things wrong with IV studies. Usually they test the addition of a substrate (for example a single amino acid) directly in the blood, through IV. This is not how we eat protein. We eat protein as a whole food and thereby trigger a set of hormones that could be of influence which are not triggered through IV. Testing whole food protein makes it more difficult to isolate protein specific effects but that is less relevant. We want to look at the effects of eating meat or other protein sources in real life.
If you checked my previous writing on the subject then you already know that incretins, which are triggered during meal ingestion, make a difference in the insulin (and maybe also glucagon?) response.
In a study, further down this article, we noted a slight increase in insulin. This increase in insulin cannot be simulated with just an IV injection of a single amino acid resulting in a failure of IV to be representative for the real world. Likewise it is with the rate of absorption of amino acids and the effect of additional fat in the meal.
IV studies also fail to account for the full labeled amino acids, only noting there is little appearing in the plasma glucose. Plasma glucose is only output from the glucose producing/releasing organs. This will be further clarified when we look at the kidneys.
Update 2020.04.12:
Further studies found clearly demonstrate the effects of incretin and how they are important for the level of secretion of glucagon and insulin. Especially the second link clearly explains the interplay between incretin and the alpha- and beta-cells in the pancreas to regulate the hormonal secretion.

“Incretin and islet hormonal responses to fat and protein ingestion in healthy men” https://journals.physiology.org/doi/full/10.1152/ajpendo.90233.2008
“Expanding the definition of the incretin axis” https://www.facebook.com/whitemcgarrahlab.dmpi.7/videos/120038229629085
Cortisol
One other such dietary effect is cortisol. Cortisol is elevated when eating a meal. This is independent of the amount of protein and is significant. For women the effect is much less pronounced.
“Influence of Consumption of a High-Protein vs. High-Carbohydrate Meal on the Physiological Cortisol and Psychological Mood Response in Men and Women”, Sofie G. Lemmens, Jurriaan M. Born, Eveline A. Martens, Mieke J. Martens, Margriet S. Westerterp-Plantenga, 2011, https://journals.plos.org/plosone/article/file?id=10.1371%2Fjournal.pone.0016826&type=printable
Glucocorticoids to which cortisol belongs appear to have a synergistic effect with glucagon on GNG meaning that together they are able to increase GNG more than each individually can. This synergy takes place in the liver and in the kidney. Both glucagon and cortisol result in an increased expression of PEPCK-C which is a rate limiting enzyme for GNG.
“Factors that control the tissue-specific transcription of the gene for phosphoenolpyruvate carboxykinase-C”, Chakravarty K, Cassuto H, Reshef L, Hanson RW, 2005, https://www.ncbi.nlm.nih.gov/pubmed/15917397
Kidney GNG
The kidneys are heavily involved in the regulation of plasma glucose. It seems they have a compensating role. I’ll first explain and then we pull up some papers.
Upon feeding, protein stimulating glucagon would normally increase glucose release from the liver, coming from both glycogenolysis and gluconeogenesis. Yet studies show no such increase in plasma glucose. There is however also an increase in insulin in normal meals. Very mildly when the protein is eaten with fat and more severely when ingested with carbohydrates. Despite the elevated insulin, we don’t see a drop in glucose but insulin is very potent at blocking glycogenolysis. The reason we don’t see this drop is because the kidneys are also stimulated in gluconeogenesis by glucagon. The kidneys increase output while the liver reduces output and builds up its glucose reserve. This is an ideal situation when feeding as it gives the liver the chance to increase its reserve!
Let’s first have a look at this interesting fact from 1964. They found that the kidneys increased their GNG in the presence of ketones and short-chain fatty acids.
“ACCELERATION OF RENAL GLUCONEOGENESIS BY KETONE BODIES AND FATTY ACIDS”, HA KREBS, RN SPEAKE, R HEMS, 1964, https://portlandpress.com/biochemj/article-abstract/94/3/712/54563/ACCELERATION-OF-RENAL-GLUCONEOGENESIS-BY-KETONE
That is really interesting as it could again indicate that the kidneys help the liver to maintain/save its glycogen reserve as much as possible by assisting in the maintenance of plasma glucose through GNG.
The following review on kidneys is very extensive and interesting in total but I want to know about the postprandial part. They came to the same conclusions when investigating postprandial GNG from the kidneys.
Seemingly, renal glucose release paradoxically increases postprandially and it accounts for >50% of the endogenous glucose release for several hours. These observations suggest that increased renal glucose release may play a role in facilitating efficient liver glycogen repletion by permitting the substantial suppression of hepatic glucose release.
“Renal Gluconeogenesis”, John E. Gerich, MD, Christian Meyer, MD, Hans J. Woerle, MD and Michael Stumvoll, MD, 2001, https://care.diabetesjournals.org/content/24/2/382#sec-13
When running the test on rats we can dissect and see what effect it had on the liver. They increase GNG, contributing for about 15%~20% of the post-absorptive endogenous glucose production (EGP, what is found in circulation). This raises blood glucose levels and causes a slight rise in insulin. Enough insulin to reduce the output of glucose by the liver so that the liver can fill up its glycogen store.
Protein feeding ameliorated the suppression of EGP by insulin and the sparing of glycogen storage in the liver but had no effect on glucose uptake… Protein feeding increases kidney gluconeogenesis without increasing global endogenous glucose production, and improves insulin suppression of the latter, likely at the hepatic site.
“Protein Feeding Promotes Redistribution of Endogenous Glucose Production to the Kidney and Potentiates Its Suppression by Insulin”, Bruno Pillot, Maud Soty, Amandine Gautier-Stein, Carine Zitoun, Gilles Mithieux, 2009, https://academic.oup.com/endo/article/150/2/616/2455542
They provided a low dose of insulin and found that with the high protein diet, the liver glycogen was significantly higher (almost 4-fold) than the normal diet.
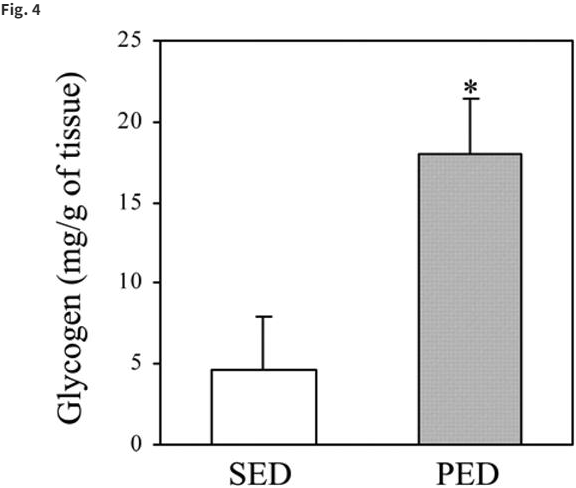
Important here is that we are talking about a rat study. They have roughly a 7x higher metabolism. In order for them to be ketogenic, protein has to be reduced to about 10%~15% depending on the addition of carbs in the diet or not, so they may be more optimized to GNG and therefore able to fill up their liver faster. The authors of the study also referred to their study on humans where they found a similar contribution of the kidney’s to glucose production.
In another rat study, we note similar results. An increase in gluconeogenesis and a reduction in glycolysis when shifting to a zero carbohydrate diet.
“Gluconeogenesis from dihydroxyacetone in rat hepatocytes during the shift from a low protein, high carbohydrate to a high protein, carbohydrate-free diet.”, Azzout B, Chanez M, Bois-Joyeux B, Peret J, 1984, https://www.ncbi.nlm.nih.gov/pubmed/6491768
Taking everything together we see that protein is converted to glucose by both the liver and kidneys. The liver reduces output to increase its glycogen reserve while the kidney provides a backup to maintain blood glucose.
The supply is used to fill up the liver glycogen reserve as much as possible. Storing it as glycogen requires first to pass through GNG.
update 2022.08.17
A review article came up with the same conclusions:
The kidney accounts for 10% systemic gluconeogenesis in the absorptive phase; the rate rises to as much as 25% in the post-absorptive phase[32]. Moreover, in the case of prolonged fasting, the kidney prevents and reverses hypoglycemia by a counter-regulatory process of increased gluconeogenesis and inhibition of glucose uptake[33]. Besides such adaptive changes, impaired renal insulin signaling/sensitivity affects renal gluconeogenesis[15]. Improving renal insulin sensitivity may reduce systemic glucose levels via gluconeogenesis inhibition [34]. In the postprandial state, the renal glucose release accounts for approximately 50% of the endogenous glucose release for several hours. These observations suggested that increased renal glucose release may play an important role in facilitating efficient liver glycogen repletion by permitting substantial suppression of hepatic glucose release. Hormones (notably insulin and catecholamines), substrates, enzymes, and glucose transporters are some of the other factors which affect glucose production by the kidney[31,35-39].
For example, a 61% decrease in overall glucose release via hepatic glycogenolysis was reported previously in a human study, virtually ceasing in 4 to 6 h[54]. This finding was attributed to the need for replenishing the liver glycogen stores and to limit postprandial hyperglycemia. Moreover, unlike the liver, renal gluconeogenesis increases by approximately two-folds and accounts for 60% of endogenous glucose release in the postprandial phase[54].
“Renal gluconeogenesis in insulin resistance: A culprit for hyperglycemia in diabetes” https://www.wjgnet.com/1948-9358/full/v12/i5/556.htm
Longevity
This is not at all backed up with research, just ideation. After reading the book on longevity from David Sinclair where he explained how cells wait for the ideal circumstances to proliferate, I thought about how that would translate to our bodies and protein ingestion (or actually food in general). We live our lives fasted most of the time. Eating breaks that situation for a brief moment and signals the body to make the best use of it. We live in plentiful times now but having food was not always obvious in our past. As such, cell proliferation is a key event and we know this by increased mTOR activity for growth, activated by both insulin and certain amino acids. However, such growth doesn’t come without an energy cost. Part of the food itself must serve as fuel and part of that food must be stored as backup for later energy requirements. In such light it makes sense that any excess is converted and put into our energy reserves which is our adipose and glycogen. The glycogen in our liver is a constantly fluctuating buffer and must be filled at times when supply is plenty. This does not happen easily under a ketogenic diet when carbohydrate sources are heavily reduced or even eliminated as on a carnivore diet.
The body can’t afford to have completely depleted glycogen in the liver. It has to keep it at a minimum level to respond to exercise activities and also in response to pathogens to regulate the immune system. As such, again, it would seem logic it would take any opportunity to refill the glycogen store as much as possible whenever there is excess.
The glycogen buffer in our liver is key for survival. As with any buffer, you refill whenever supply is available.
Type 1 Diabetes (T1D)
Update 24 April 2020: fully added the T1D section
To demonstrate the higher GNG rate we need to disable insulin because, as you could read above, insulin causes a reduction in output of glucose from the liver to build up the glycogen.
If GNG would be demand driven then insulin would not make a difference. You could say it makes a difference for glycogenolysis so to rule this out we can look at T1D who do not produce (or very little) insulin. Their fasting insulin and post-meal insulin is pretty much the same so there would be no difference in glucose output from the liver when comparing high versus low protein content in the meal.
But it does, the next reference tested exactly this point. When the protein content in a meal is higher, T1D need to adjust for it by increasing their insulin. There is not a single study that shows that protein cause a greater glycogenolysis so where is the extra glucose coming from? Indeed, from the glucogenic amino acids through the GNG process.
The 60 gram of protein versus the 5 gram of protein resulted in a 50% greater need for insulin. Carbs and fat were kept the same.
“Dietary protein affects both the dose and pattern of insulin delivery required to achieve postprandial euglycaemia in Type 1 diabetes: a randomized trial” https://onlinelibrary.wiley.com/doi/abs/10.1111/dme.13875
Amino Acids directly stimulate glycogen synthesis (added 2021.01.21)
Another study found that amino acid availability induces glycogen synthesis. This happens through inhibition of glycogen synthase kinase 3 (GSK-3). It was shown in muscle cells which does not guarantee it also happens in the liver…
“Regulation of glycogen synthesis by amino acids in cultured human muscle cells” https://pubmed.ncbi.nlm.nih.gov/11013237/
But it does.. A study was done by inhibiting GSK-3 in diabetic rats. They found that the inhibition caused a 3-fold increase in liver glycogen synthesis and lowered plasma glucose.
“Effects of a novel glycogen synthase kinase-3 inhibitor on insulin-stimulated glucose metabolism in Zucker diabetic fatty (fa/fa) rats” https://pubmed.ncbi.nlm.nih.gov/12351425/
Not only amino acids inhibit GSK-3, insulin does this as well and we have seen that dietary protein also induce an insulin response.
“Glycogen Synthase Kinase-3 in Insulin and Wnt Signalling : a Double-edged Sword?” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4485494/
Finally
It looks like both glucagon and insulin are active on the liver at the same time. Glucagon triggers glycogen breakdown and GNG, 2 sources to release glucose into the bloodstream. Insulin however opposes the release, the glycogenolysis and causes the generated glucose to be stored in the liver.
During this phase, the kidneys cover for some of the effect by increasing its glucose production.
“Plasma Glucagon and Insulin Responses Depend on the Rate of Appearance of Amino Acids after Ingestion of Different Protein Solutions in Humans”, Jose A. L. Calbet, Dave A. MacLean, 2002, https://academic.oup.com/jn/article/132/8/2174/4687709
The degree to which insulin opposes the situation will dependent on the level of amino acids that is reached in the plasma but the rise in insulin noted in the different experiments show it is sufficient to increase the liver glycogen levels.
As such, if you are concerned about ketone production you have to properly balance out protein with your activity level.
Being very active in a way that demands extra glucose will pull glucose from the liver glycogen store during activity and afterwards to replenish muscle glycogen store.
Not being active you need to temper your protein intake. Your demand would be lower anyway as you don’t have as much muscle repair and growth going on.
How much protein? As always, measure your ketones to know how much you are producing. It’s individual, nobody can tell for you personally where the tolerance level is and then it will still depend on different situations, varying day by day.
— The End —
Leave a comment